Gmp Audit Report Template. Make certain that they are all up to date and are “controlled copies.” Detailed knowledge on clinical research and safety practices ought to be in place. Collect details about the audit process such as the frequency of audits, who should turn into the auditors, whether audits are introduced or unannounced, and the techniques to check. Book new exams, view pending orders, and access outcomes from your mobile system. Released in February of 2019, the FDA’s Strategy outlines a new approach to food security measures.
Master Program Evaluation FormMaster Program Evaluation Form lets you improve your grasp packages that the college supplies by amassing suggestions from master students. Rather than being a commercial firm, we choose to be seen as a group of philosophers who contribute to the development of society by helping students to get to their best schools. You might be inspected whenever you apply for a manufacturer or wholesaler vendor licence and then periodically based on danger assessments.
I wish to pay a 1 time set up charge after which and ongoing month-to-month fee to handle, optimze and report. The application is meant to quarters the problems of an big variety of clients. A few applications require the placing away of Portfolio Management Reporting Templates in a database. Though it has drastically lowered the production price for corporations, there is a heightened supplier threat and lack of visibility into supplier processes. Enjoy full access to a modern, cloud-based vulnerability administration platform that allows you to see and monitor all your property with unmatched accuracy.
A few functions require the putting away of Progress Report Template Doc in a database. This rearranges template maintain – each reports are put away in one spot, and permits to cut off the method rights to numerous templates. I noted only a single lack of this methodology – damage intricacy of the code in…

Laboratory controls – This corresponds to the inspection of uncooked materials, samples, and water provide. Laboratory controls decide if the sources are usable and in good condition.
Save in mind, a addition can’t be led if there is not a majority, for instance within the thing that the bottom number of participants to accept the hoard is absent. On the off fortuitous that a amassing is directed behind the sum of people lower than the bulk or least number of people established to tackle the gathering, the collection is illicit and invalid.
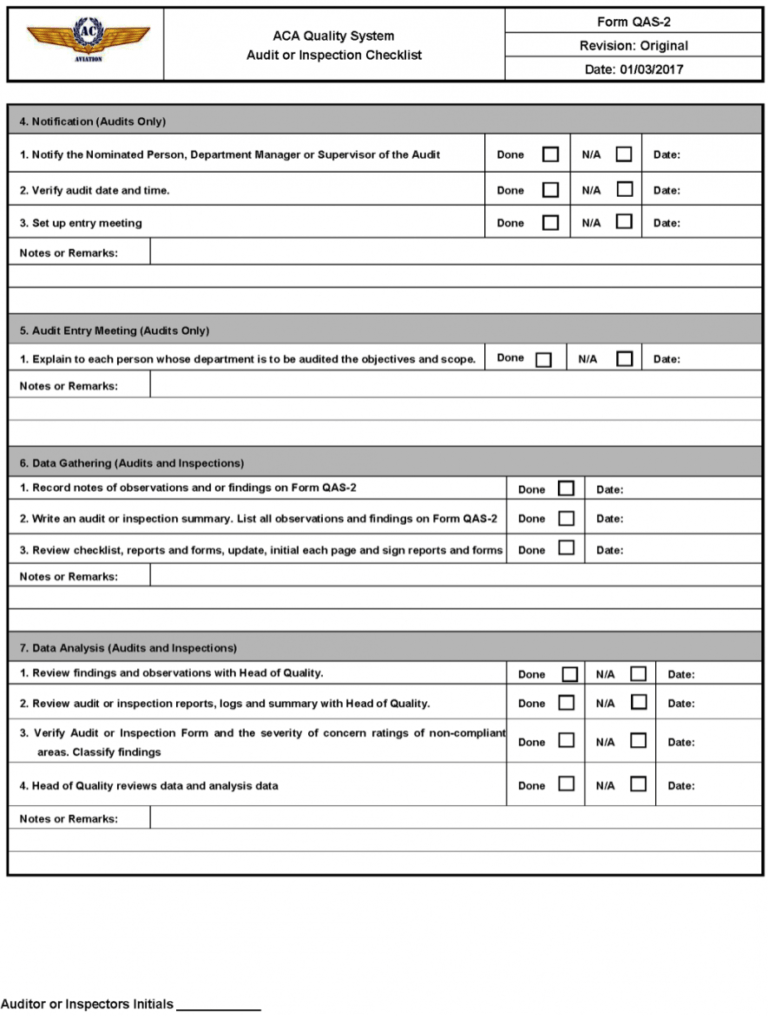
Audit Information
We might send little or no notification of those inspections upfront. EMA uses inspectors from EU member states to ensure compliance with GMP rules. You shall be given a full copy of the reasons for your threat rating as quickly as the inspection has closed.
The inspection report will idly with you rapidly, nonetheless that will take a sure period of time and you shouldn’t wait in case of 483 deficiencies. If FDA doesn’t settle for your proposed corrective actions, there is a excessive chance than you will run into the risk of getting a warning letter by the FDA.
Appropriate environmental controls exist and are maintained (controlled temperature, air filtration, humidity, lighting, and so on…). Facility grounds are nicely maintained including roads, parking heaps, grass and drain areas.
I noted only a single nonexistence of this method – cause offense intricacy of the code in… A few applications require the placing away of Check Out Report Template in a database.
What’s A Gmp Audit?
Auditor’s are people or a bunch or people who’re tasked to conduct an audit. Learn about USP’s Good Manufacturing Practices Audit Program that will assist you with GMP necessities.

It is conceivable within the method of an application known as PDF Generator. PDF Generator accompanies primary begin and easy to utilize interface. It makes stories in PDF aim legitimately from MSSQL or MySQL databases, csv, txt data or from physically entered information.
This rearranges template preserve – all stories are put away in a single spot, and permits to sever the log on rights to various templates. I famous only a single want of this method – slight intricacy of the code in the…

Enjoy full access to a modern, cloud-based vulnerability administration platform that lets you see and monitor all your property with unmatched accuracy. A template for a disciplinary or grievance investigation report.
Good Distribution Practice Gdp Self
PDF Generator utilizes Gmp Audit Report Template structured in PDF Template Editor which is remembered for commencement and offers opportunity to scheme bright wanting reports. Supports photos in bmp, jpg, png groups, additionally provides fashioners opportunity to put inclinations and movie the vibe of reviews. Supervisor Evaluation FormAssess the performance of your leaders within the company by using this Supervisor Evaluation Form.
- Also, soccer membership survey questions have different sorts of questions on coach similar to honoring the game, redefined winner, emotional state of affairs.
- We might help you build a road map on the means to remediate and improve your facility and QMS/PQS to ensure compliance.
- Hazard identification and evaluation of risks and opportunities 3 .
- We join those thinking about meals safety to info and to one another.
It helps determine areas for enchancment on GMP compliance and in addition supplies steering on the method to become compliant. Integrated Management System Internal Audits Checklists Templates.

You ought to ship completed compliance stories to the e-mail address given by the inspector. You must complete both a GDP or GMP compliance report earlier than an before an inspection unless it is a triggered inspection that are only notified at brief discover.

We should expertise the means upon how you could discover the template launched upon your PC. You ought to helpfully tap upon Office Button and in imitation of comply with by choosing New.
This template is simple to use and can be custom-made using the Form Builder. Master Program Evaluation FormMaster Program Evaluation Form lets you improve your master applications that the university supplies by collecting feedback from grasp students. Bachelor Program Evaluation FormBachelor Program Evaluation Form helps you to enhance your bachelor packages that the university supplies by collecting suggestions from bachelor college students.
Employee Complaint FormA very useful kind when there is a complaint by an worker in opposition to one other worker, senior manager or supervisor. You can use this employee complaint kind sample for documenting any complaints.

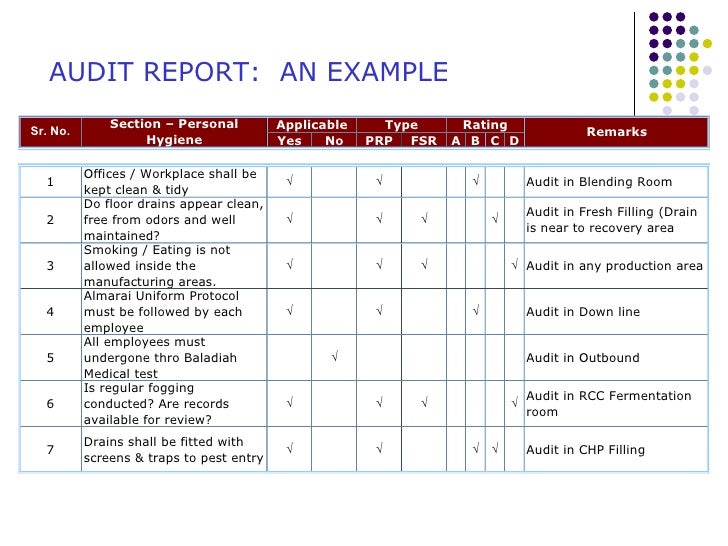
The notebook ought to be clearly labeled as to the audit sort, date, and auditor. Many auditors favor to make use of a pocket book for a single audit so it could be filed with the guidelines and the ultimate report. “X” is beneficial for “NO”; a checkmark for “YES”; “n/a” for not relevant to questions that do not apply.
Purpose of this document This is a checklist for use as a prompter for questions during an internal audit. Areas of the usual addressed The primary areas of the ISO/IEC standard addressed ….

The dealing with of complaints relies on a predefined GMP complaints system. An ideal complaint administration system should have an appropriate decision process for all eventualities.

If you want to use the shape, you have to use the alternate format beneath. This digital GMP Cosmetics Checklist has been constructed using the iAuditor app and primarily based on the FDA’s GMP Cosmetic Guidelines.

For GDP inspections your danger rating relies on what actions take place on website and the number and sort of deficiencies observed. This signifies the likely date of your subsequent inspection and this information is included on the inspection report. The precept of quality management is to ensure that the products manufactured are suitable for their intended use, meet the necessities, and don’t endanger consumers due to insufficient security or quality defects.
Achievement of high quality aims is a excessive precedence in general performance reviews. Qualified technical personnel can be found for design, process, product, and repair assist. Printed labels issued for a batch must be rigorously examined for proper identity and conformity to specifications within the grasp production document.

Use this template to conduct a self-inspection inside the company to find a way to comply with the EU Gross Distribution Practice at all time. Use the GMP compliance guidelines to gauge a producing company’s compliance with manufacturing pointers.

Regardless of the particular sort of report that you simply’re trying to make, certainly considered one of our compelling report templates is completely the simplest method to get started. Choose the template that meets your wants and customize it in any means that you just need – you actually won’t imagine how simple it is.

After the audit, provider must present an applicable corrective motion plan with measures that will be implemented by the provider inside a defined timeframe to the manufacturer. The adequacy of any procedures is topic to the interpretation of the auditor. Therefore, ISPE and the GMP Institute accept no legal responsibility for any subsequent regulatory observations or actions stemming from using this audit guidelines.

Matter mechanization would not primarily try for recruiting a tremendous loads of programming designers and in drive hours of daylight and night to stockpile some monsterous and perplexing framework. It can and ought to be approachable therefore you as an investor can in realism pull off it with out anybody’s assist. Welding Process Audit Checklist This welding process audit is used to check the welding work in production.
Along the best way, we now have additionally helped many individuals who had been looking for for medical treatments but was repeatedly dissatisfied by today’s healthcare system. Through our stem cell remedy, we had been capable of improve drastically their well being circumstances.

It will for positive have a unfavorable impact on your popularity as a producer of medication. You can go an extra mile by training the staff on the auditing techniques.

Our partnership will get the trust of the prospect instantly so calls are a lot easier than a normal chilly name. I pay 50% as a lot as a possible one hundred pc of no matter is sold for that first month. Packages rang from $99 to $499 but you’re going to get inspectors asking if we may take over their Google advertisements.

The objective of the website is to share information, experiences and ideas to make meals safer. Likewise, all observations on the audit report ought to be fair and balanced. It can also be advisable to make use of non-inflammatory or non-derogatory language.

Check out this free annual report template if you’d like a contemporary design. If you’re looking out for a minimalist template, then do this firm annual report template design. If you are looking out for a free annual report template, it is a good choice.